ASGCT Q1 Landscape Report Paints Positive Picture for Gene and RNA Therapy
BOSTON – The CEO of the American Society for Gene and Cell Therapy (ASGCT), David Barrett, JD, presented highlights from the Society’s latest Landscape Report on Cell, Gene and RNA Therapy for the first quarter (Q1) of 2026.
The ASGCT report is developed in conjunction with Citeline, a subsidiary of Norstella (a pharmaceutical intelligence provider covering drug development from preclinical to commercialization).
Barrett said there are currently 42 gene therapies approved worldwide, along with 38 RNA therapies and 76 (non-genetically modified) cell therapies, which are steadily growing the field. Two cell therapies were approved in Japan in Q1.
There was a small increase in deal-making, and a significant 30% increase in startup funding compared to the same period in 2025. “I think that signals and underscores a rebounding sector,” said Barrett.
Of the eight gene therapies approved over the past 12 months, half were in the United States, with three more in China. “The regulatory pace is starting to pick up, another strong indicator for the future of our field,” Barrett said. It is a similar picture in RNA therapies. “We see a steady uptick over the course of the last year,” he added.
Zooming out, Barrett estimated that there are more than 4,200 therapies currently in development, from preclinical through pre-registration. The vast majority of those (more than 4,130) are gene and genetically modified cell therapies, including about 1,300 RNA therapies.
In the field of gene-modified cell therapies, CAR T continues to lead the pipeline for ex vivo gene therapies, with natural killer (NK) and T-cell receptors gaining traction. Not surprisingly, genetically modified cell therapy overwhelmingly targets cancers, but Barrett noted growth in the percentage of these therapies targeting immunological diseases, including lupus, multiple sclerosis, and HIV.
Pipeline growth
Barrett also noted growth and “a promising future” in the clinical trials pipeline. There are currently 350 Phase I, 319 Phase II, and 41 Phase III trials in gene therapy (up from 35 a year ago). “Hopefully, we will see a number of completed trials and FDA decisions in the near term,” said Barrett. A growing proportion of gene therapy trials (exceeding 60 percent) is for non-oncology indications.
In the RNA space, “RNAi therapies are jumping,” said Barrett. The same cannot be said, however, for mRNA. “Unsurprisingly, mRNA therapies continue to slide quarter over quarter,” a symptom of “shaken confidence” in that space, he continued. RNA therapies are targeting primarily non-oncology indications, especially in rare diseases.
Upcoming catalysts
On the business front, Barrett noted there has been “a nice uptick” in Q1 in start-up funding compared to the same quarter last year, which he deemed “a really promising indication.” The number of start-ups historically has tended to hover between 5-20. For Q1, that number was 26.
The Q1 report tracks various business catalysts anticipated through the end of 2027, including increased interest and uptake in expedited review designations—fast track, RMAT, orphan drug breakthroughs and other accelerated approval pathways.
“FDA is getting a lot done… and hopefully we’ll see the same moving forward,” Barrett said.
The full Landscape Report is available online from the ASGCT website.
The post ASGCT Q1 Landscape Report Paints Positive Picture for Gene and RNA Therapy appeared first on GEN – Genetic Engineering and Biotechnology News.
ASGCT 2026: Beverly Davidson Offers Vehicle and Route for Huntington’s Disease Gene Therapy
BOSTON – Geneticist Beverly Davidson, PhD, received the 2026 Outstanding Achievement Award from the American Society of Gene and Cell Therapy (ASGCT). Davidson is currently the chief scientific strategy officer at the Children’s Hospital of Philadelphia (CHOP) and a former president of ASGCT.
Some of the research Davidson presented was conducted at a new biotech company she co-founded called Latus Bio, which earlier this month announced it had raised $97 million in a Series A round. The company develops novel AAVs to specifically target central nervous system (CNS) disorders, with a lead program in Huntington’s disease (HD).
After thanking her mentors—Bill Kelly, MD, Michael Welsh, MD, and Kathy High, MD—Davidson turned her attention to presenting new advances in engineered gene therapies. Throughout her career, she has focused on improving adeno-associated viruses (AAVs) for CNS gene therapies, with a particular emphasis now on HD. Key elements include selecting the right cargo and developing the appropriate delivery vehicle. Her goal is to scale lab research in neurons, mouse models, and non-human primates (NHPs) to treat patients, including adults with HD.
Major hurdles to tackling genetic diseases of the brain include scalability and a lack of potency, Davidson said. The search for alternative AAV serotypes to AAV2 that could target neuronal cells began back in 2000. IV administration does not provide sufficient targeting to the brain. Even AAVs that have been engineered to enter the brain from the blood have high peripheral exposure and a high cost of goods per patient, which significantly lowers scalability and impact. (In one study, liver biodistribution of AAV was many orders of magnitude higher than in the CNS.)
Davidson focused on HD, the late-onset, dominantly inherited genetic disease. The identification of the gene harboring the HD mutation in the early 1990s by a consortium of researchers was one of the biggest success stories in human genetics. Even more remarkable was the underlying disease mechanism—the expansion in exon 1 of the gene of a triplet repeat sequence (CAG) producing an abnormally long string of glutamine residues in the huntingtin protein.
The right target
One of the major challenges in devising a gene therapy for HD is ensuring that the therapeutic reaches the right network—the deep brain and cortical areas. Therapies have to reach the right circuit, and the right cells in those circuits, Davidson said. Over the years, her group has tailored AAVs for delivery to the brain, inserting peptides into exposed loops of the virion to allow for targeting and unbiased diversity for blood-to-brain delivery. Nowadays, she said, machine learning approaches can be applied for further capsid improvements.
Davidson’s CHOP lab developed a method for screening AAVs with enhanced potency for CNS therapies. After generating huge libraries containing tens of millions of novel capsids, the group performed serial enrichments to identify the most attractive capsids. After screening pools of injected capsids into two species of monkeys, a winning capsid emerged: AAV-DB-3.
Davidson’s group infused AAV-DB-3 into NHPs, looking for targeting to the putamen (base of the forebrain) and caudate regions. Those results were published in Nature Communications in 2025. “AAV-DB-3 really stood out for its ability to transduce deep layer cortical neurons that are important” in HD, Davidson said. Moreover, the results were achieved with relatively low doses and only required a single infusion per hemisphere, outperforming the widely used AAV5.
Somatic instability
With a promising delivery vehicle identified, Davidson next addressed the therapeutic strategy, which takes aim at the somatic expansion of the CAG repeat. This codon grows longer over time in certain cells in the brain, sometimes expanding to hundreds of repeats.
MSH3 is a DNA repair protein that is required for CAG repeat expansions, as seen in mouse models of HD and other triplet repeat disorders, including myotonic dystrophy. Research led by Paul Ranum, PhD, who is a co-founder of Latus Bio, posted in a preprint on bioRxiv earlier this year, modeled the impact of lowering levels of MSH3 on somatic instability.
Ranum and colleagues used an artificial microRNA showed to lower MSH3 levels in NHPs by 48-94 percent. Computational modeling suggests that this would reduce somatic instability and delay onset of HD symptoms by many years. Early studies using a well-known HD mouse model, the Q111 mouse, to assess biodistribution, quantify knockdowns, and assess the impact on somatic CAG repeat expansion. AAV-DB-3 expression is highest in the striatum and cortex at 16 weeks, dropping MSH3 levels by 50%.
Davidson closed by emphasizing the need to ensure scalability for treatment beyond ultra-rare disorders. Latus hopes to file an Investigational New Drug application for its HD therapy, LTS-201, in the second half of 2026. At least two other biotech companies are also targeting MSH3 by other means.
The post ASGCT 2026: Beverly Davidson Offers Vehicle and Route for Huntington’s Disease Gene Therapy appeared first on GEN – Genetic Engineering and Biotechnology News.
ASGCT 2026: AI-Optimized Cas12l Gene Editor Offers Compact Cas9 Alternative
BOSTON — In a potentially significant advance for the genome editing field, researchers from the biotechnology company Caszyme and the Vilnius University Institute of Biotechnology in Lithuania have developed a potent and compact variant of Cas12l nuclease. Giedrius Gasiūnas, PhD, Caszyme co-founder and CEO, presented highlights of the research at ASGCT.
The work represents “a great example of the potential of continued mining for novel Cas effectors within the bacterial metagenomic diversity dark matter,” said Rodolphe Barrangou, PhD, Editor in Chief of The CRISPR Journal, which will shortly be publishing a paper on the Lithuanian team’s results.
“We need more diverse effectors to address the technical shortcomings of the CRISPR toolbox,” Barrangou continued. “This study is a great illustration of the potential of mining bacterial diversity.”

The Lithuanian team, including veteran gene editor Virginijus Siksnys, PhD—winner of the 2018 Kavli Prize with Jennifer Doudna, PhD, and Emmanuelle Charpentier, PhD, for CRISPR gene editing—used a hybrid approach to optimize Cas12l. By combining cryo-electron microscopy (cryo-EM) structure-guided design with artificial intelligence (AI) protein language models, the team was able to engineer a variant (Asp2Cas12l M82) that overcomes the known efficiency limitations of the Cas12l family.
Although Cas9 has widespread utility, including clinical applications, researchers have long considered its relatively large size and requirement for G-rich protospacer adjacent motifs (PAMs) problematic. The Cas12l family, discovered in the Armatimonadota bacterial phylum, offers a more compact size (867 amino acids) and recognition of a C-rich PAM site.
But wild-type Cas12l enzymes exhibit lower editing efficiencies and higher target-to-target variation compared to Cas9. According to Gasiūnas, the new M82 variant is “reliable, precise and adaptable,” and shows promise for a wide range of therapeutic applications.
“Through our continued work exploring novel Cas systems, Caszyme is focused on advancing technologies that move beyond promise into practical use.”
Path to potency
The engineering of the M82 variant proceeded in two steps. First, the Caszyme researchers solved the 3D structure of Asp2Cas12l complexed with an sgRNA and DNA to high resolution (2.51 Å). This revealed a unique “bracelet” architecture whereby the nuclease encircles the DNA target via interlocking helical bundles and a proline-rich string.
Next, the team introduced arginine substitutions at dozens of positions in the molecule to enhance electrostatic attraction to the negatively charged DNA backbone. This work included the production of an M67 variant, which provided a 7-fold improvement in indel editing over the wild-type nuclease.
To engineer further refinements, the Caszyme group turned to AI, specifically the ESM-2 protein large language model. This model predicted evolutionary hotspots considered likely to preserve or enhance function. Integrating these AI-derived substitutions—Q572R in the bridge helix and F607S in the RuvC domain—resulted in the final M82 Cas12l variant, illustrating the value of AI-supported engineering rather than deploying protein-directed evolution.
Rivaling Cas9
Gasiūnas presented data showing that M82 possesses good activity across recalcitrant gene targets, reducing the target-to-target variation that plagues many novel nucleases. In head-to-head comparisons in HEK293T cells, M82 demonstrated an average indel editing rate of 67.4%, nearly identical to that of Cas9 at overlapping target sites. This potency was consistently maintained across several delivery formats, including plasmid DNA, mRNA, and ribonucleoprotein complexes.
The Caszyme group also showed excellent M82 efficiency in homology-directed repair (HDR). In experiments targeting the AAVS1 locus, M82 facilitated a site-specific gene insertion frequency of 39%, outperforming Cas9 in the same context. Using single-stranded donor templates, HDR rates reached as high as 56%. Gasiūnas suggested that the staggered cut produced by Cas12l may inherently steer DNA repair toward precise correction rather than stochastic indels. With regard to safety, Caszyme found that M82 Cas12l maintained a high degree of on-target precision. Secondary editing signals were largely detected at or near the lower limits of assay sensitivity, suggesting a low risk of off-target cleavage.
The compact size of the M82 variant makes it an attractive candidate for adeno-associated virus-mediated delivery, which has strict limits on cargo size. “It is no secret that the CRISPR space has faced challenges and concerns in recent years,” Gasiūnas said. “However, we are confident in M82’s ability to create headroom for scientists to stand up and innovate within.”
Crowded field
Cas12l is not the only compact Cas nuclease gaining attention, of course. In a talk preceding Gasiūnas’ presentation, Zhaoshi Wu, PhD, co-founder and chief technology officer of Shanghai-based Castalysis Bioscience, presented an update on Cas12n, details of which were first published in Molecular Cell in 2023. The nuclease was touted as being the first independent CRISPR-Cas complete gene family uncovered by Chinese scientists within China’s territory.
Touted as a next-gen ultra-compact gene editing system, Cas12n (branded as alphaCas) consists of just 450 amino acids, and possesses structural similarity to TnpB. Cryo-EM structural analysis led the Chinese investigators to optimize the molecule for non-viral in vivo delivery. Preclinical experiments showed robust genome editing in a mouse model by targeting PCSK9 using lipid nanoparticle delivery, resulting in sharp drop in serum LDL levels.
Wu said his company is on target to begin its first clinical before the end of 2026. But he faced an uncomfortable moment during audience questions. Fyodor Urnov, PhD, challenged Wu’s claim that an inherent advantage of Cas12n was its safety profile compared to Cas9. Urnov pointed out that Intellia Therapeutics has two ongoing Phase III in vivo trials using CRISPR-Cas9 that show no immunogenicity concerns using LNP delivery.
Urnov later congratulated Wu on the rest of the company’s data and wished them success.
The post ASGCT 2026: AI-Optimized Cas12l Gene Editor Offers Compact Cas9 Alternative appeared first on GEN – Genetic Engineering and Biotechnology News.
ASGCT 2026: Rare Instance of AAV Integration into Human Genome Linked to Brain Tumor

BOSTON — A team at Children’s Hospital of Philadelphia (CHOP) led by Rebecca Ahrens-Niklas, MD, PhD, and Lindsey George, MD, has described a case of a brain tumor linked to a rare integration of adeno-associated virus (AAV).
George presented the work at the American Society of Gene and Cell Therapy (ASGCT) conference in a plenary talk selected as the “presidential abstract” by ASGCT president, Terry Flotte, MD. The study, “Neuroepithelial tumor with AAV integration after intracisternal magna vector delivery,” was published in the New England Journal of Medicine.

Over the past 25 years, some 6,000 patients have been treated with some form of AAV gene therapy. In all that time, George said, there have been no established long-term safety concerns, although genome integration events have been reported in mouse and dog studies. But the case documented by George and colleagues at CHOP suggests that the gene therapy field may need to pay more attention to this potential occurrence.
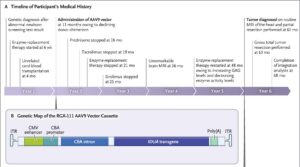
The story began with a 5-year-old boy with an inherited lysosomal disorder, severe MPS1 deficiency (Hurler subtype). The patient received enzyme replacement therapy at six weeks of age, followed by a cord blood stem cell transplant at age four months.
Investigators chose to perform gene therapy when the patient was 13 months old to deliver the iduronidase (IDUA) gene. The vector chosen was an AAV9 serotype, using a cytomegalovirus enhancer and a chicken beta-actin promoter driving the gene. The virus was administered into the boy’s cisterna magna in the base of the skull.
When the boy was five years old, a routine neurological scan revealed a large intraventricular mass that had not been observed two years earlier. Analysis of the tumor revealed it was a PLAG1-driven neuroepithelial tumor—indeed, PLAG1 expression was almost 300 times higher than in other central nervous system tumors studied at CHOP. (PLAG1 is usually only expressed during embryogenesis.)
Surgery to remove the tumor was successful. Eight months after surgery, there are no signs of tumor growth. The boy is also showing advanced neurocognitive function.
Tumor typing
George described RNA sequencing of the tumor, which revealed the fusion of a fragment of the AAV9 vector cassette to exon 5 of the PLAG1 gene on chromosome 8. The resulting transcript is predicted to encode a PLAG1 derivative containing five zinc-finger DNA-binding domains and a C-terminal transcriptional activation domain, which was previously reported to function as a transcriptional activator.
Curiously, the chimeric junction also included a segment of human chromosome 10, which George suspects originated during the vector manufacturing process. The integration event was present in about 40% of the total reads, suggesting integration into one of the two PLAG1 alleles.
George concluded her talk by noting that while the clinical outcome in this patient is so far encouraging, this is evidence that AAV integration can be associated with oncogenesis. The study underscores the need to monitor the most heavily transduced tissues after AAV gene therapy.
While the gene therapy community should be cautious in extrapolating this single case report across all AAV gene therapy programs, George said the study supports the use of the lowest feasible vector dose as well as tissue-specific promoters.
George noted that detection of the integrated AAV vector DNA was challenging, in part because of rearrangements of vector DNA. The use of several complementary techniques—long-read DNA sequencing, targeted PCR amplification, and RNA sequencing—was required to confirm the integration.
George and coworkers closed their paper, noting that, “Our findings support the hypothesis that rare AAV integration can contribute to human oncogenesis, which emphasizes the need to optimize gene delivery methods and monitor transduced tissues after treatment.”
The post ASGCT 2026: Rare Instance of AAV Integration into Human Genome Linked to Brain Tumor appeared first on GEN – Genetic Engineering and Biotechnology News.
Diaverum launches AI-powered kidney disease education tool
The Download: China’s AI drama factory and the WHO’s missing health targets
This is today’s edition of The Download, our weekday newsletter that provides a daily dose of what’s going on in the world of technology.
How Chinese short dramas became AI content machines
China’s short drama industry is fueled by bite-sized, melodramatic, and smutty shows built for smartphone scrolling. Now, many are being made entirely with AI: no actors, camera operators, cinematographers, or CGI specialists required.
An average of 470 AI-generated short dramas were released every day in January. Production timelines have shrunk from months to weeks, while costs have dropped by up to 90%. Storytelling is also increasingly driven by performance data.
The format is rapidly expanding overseas while reshaping the work of writers and production crews. Read the full story on AI’s dramatic impact on China’s short drama industry.
—Caiwei Chen
The world is on track to miss its health targets
The World Health Organization’s latest global statistics report reads less like a progress update than a warning sign. Progress on some of the world’s biggest health threats is stalling, and in some cases reversing altogether.
There were 1.3 million new HIV cases in 2024, malaria is resurging, vaccination rates are slipping in the Americas, and 42.8 million children are suffering from severe malnutrition. The world is now far off track from meeting many of the UN’s major health goals by 2030.
Here’s what the numbers reveal about the state of global health.
—Jessica Hamzelou
This story is from The Checkup, our weekly newsletter giving you the inside track on all things biotech. Sign up to receive it in your inbox every Thursday.
The must-reads
I’ve combed the internet to find you today’s most fun/important/scary/fascinating stories about technology.
1 As their trial goes to the jury, Musk and Altman face lying accusations
Lawyers hammered the rivals’ credibility in their closing arguments. (WSJ $)
+ Musk was accused of “selective amnesia.” (Reuters $)
+ The pair are in court over OpenAI’s future. (MIT Technology Review)
+ And their trial has made everyone look bad. (Wired $)
2 AI data centers are straining America’s power grid
Nevada is redirecting electricity from Lake Tahoe to AI. (Ars Technica)
+ Utah is getting a giant data center despite water shortage fears. (Guardian)
+ No one wants a data center in their backyard. (MIT Technology Review)
3 OpenAI is mulling legal action against Apple over its ChatGPT integration
It hasn’t got the expected benefits from its deal with Apple. (Bloomberg $)
+ OpenAI is frustrated by the promotion of the ChatGPT integration. (NYT $)
4 Anthropic has agreed terms for a $30 billion funding deal
At a $900 billion valuation, which leapfrogs OpenAI’s. (The Information $)
+ Dragoneer, Greenoaks, Sequoia, and Altimeter are leading the round. (FT $)
6 Washington and Beijing will hold formal talks on AI safety
They’ll discuss guardrails on AI. (CNBC)
+ And a protocol to stop nonstate actors getting powerful models. (NYT $)
5 Alphabet and Amazon are using “unprecedented” borrowing to fund AI
They’re tapping the foreign debt market at new levels. (FT $)
+ People can’t agree on what the AI bubble is. (MIT Technology Review)
7 Big Tech has turned to Sesame Street to deflect scrutiny of screen use
Sparking accusations of encouraging children’s tech dependence. (Reuters $)
8 Anthropic’s feud with the White House threatens other businesses
Figma and Tenable say it will harm their ability to sell software. (Bloomberg $)
9 Autonomous agents staged a digital crime spree during a safety test
The “AI Bonnie and Clyde” then deleted themselves. (Guardian)
10 A poop app analysis app offered to sell photos of users’ stools
The images were used for AI training. (404 Media)
Quote of the day
“It’s like we don’t exist.”
—Danielle Hughes, North Lake Tahoe resident and CEO of Tahoe Spark, tells Fortune that residents are being sidelined as their energy supplier prioritizes data centers.
One More Thing

The rise of the tech ethics congregation
Just before Christmas, a pastor preached a gospel of morals over money to several hundred members of his flock. But the preacher wasn’t religious, and his congregation wasn’t a church. It was All Tech Is Human, a nonprofit devoted to ethics and responsibility in tech.
Founded in 2018, the organization has built a fast-expanding community for people who believe technology should focus less on profits and more on the public interest. It’s also drawing people searching for meaning and connection in a digital world.
Find out why thousands of people are turning to tech ethics communities for guidance and connection.
—Greg M. Epstein
We can still have nice things
A place for comfort, fun, and distraction to brighten up your day. (Got any ideas? Drop me a line.)
+ Go behind the scenes of the new Lucas Museum of Narrative Art.
+ Marvel at this robot folding and launching paper planes as quickly as possible.
+ Watch the moving moments rescued animals reunite with the humans who saved them.
+ Peer into the heart of a barred spiral galaxy in this stunning new capture from the James Webb Space Telescope.

